|
Each unique substance in the chemical reaction is sunder by a plus sign (+). Assign oxidation states to all atoms in the reactants and the products (see Section 3.5 ) and determine which atoms change oxidation state. 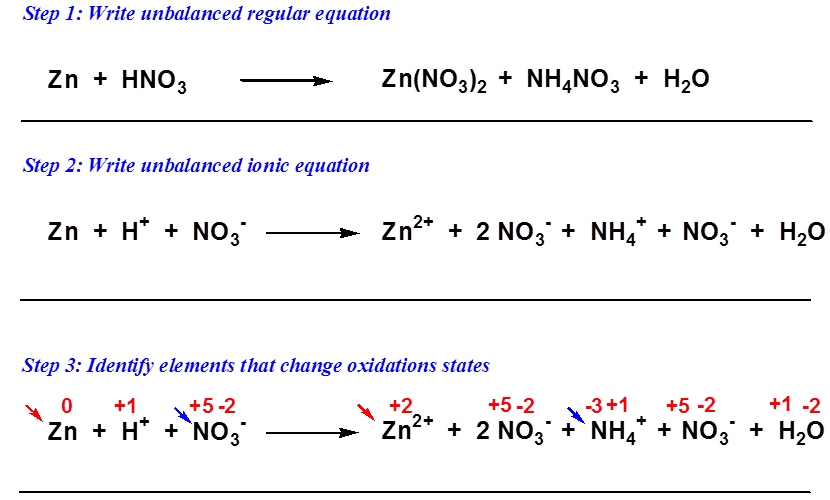
find the charge on ions (both monatomic and polyatomic. Write the unbalanced chemical equation for the reaction, showing the reactants and the products. Coefficients are whole number multipliers. When balancing equations, you never change subscripts. To be successful, you need to understand how to: balance molecular equations by changing the coefficients. Add Coefficients To Balance Mass in a Chemical Equation. Some people prefer to balance the starting reaction. Either method produces the same net ionic equation in the end. This way, when you cross out spectator ions at Step 3, you cross out equivalent numbers of ions. If you want, you can balance the equation for mass and charge first (at Step 1). 
Net ionic equations tell us what actually reacted and what we can ignore (called spectator ions). Balance the net reaction for mass and charge. The reactants and products are sunders by arrow symbols. We write net ionic equations to understand what is changing in a chemical reaction. Break up all the (aq) compounds into its ions (this is TOTAL)3. The input equation should be in the following formatĪ chemical equation is interpreted as the symbolic representation of the chemical reaction where the reactants are written on the left side and the products are written on the right side. How to write total and net ionic equations.1. Method to use the Ionic net equation calculator is as follows:ġ: Enter the chemical equation in the “Enter the chemical equation” field.Ģ: Now click the button “Balance” to get the equalize equation.ģ: Finally, for the specified chemical equation, a window will pop with the output. Find more Chemistry widgets in WolframAlpha. Steps To Balance Ionic Equations Write the net ionic equation for the unbalanced reaction. How to use Net Ionic Equation Calculator? Get the free 'NET IONIC EQUATION CALCULATOR' widget for your website, blog, Wordpress, Blogger, or iGoogle. The balanced net ionic equation calculator tool makes the prediction quick and easier and displays the answer in a fraction of seconds. 
When a chemical equation is balanced it means that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides.Net ionic and ionic equation calculator is a free online tool that shows the structure, equilibrium constant, balanced equation, substance properties with chemical formulas and names. In other words, the net ionic equation applies to reactions that are strong electrolytes in. The net ionic equation is commonly used in acid-base neutralization reactions, double displacement reactions, and redox reactions. Mass and charge must be equal in net ionic equations. The net ionic equation is a chemical equation for a reaction that lists only those species participating in the reaction. The balanced equation will be calculated along with the solubility states, complete ionic equation, net ionic equation, spectator ions and precipitates. Redox reactions, double replacement reactions, and acid-base neutralizations are where they are most frequently used. \): Regardless of the absolute number of molecules involved, the ratios between numbers of molecules of each species that react (the reactants) and molecules of each species that form (the products) are the same and are given in the chemical equation. What is Net Ionic Equation Net ionic equations are a crucial part of chemistry since they primarily reflect the things that change in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
